Carbon dioxide (CO2) is known as a greenhouse gas and plays an essential role in climate change; it is no wonder scientists have been looking for solutions to prevent its release in the environment. However, as a cheap, readily available and non-toxic carbon source, in the past few years there have been efforts to turn carbon dioxide into valuable wares, or ‘value-added’ products.
Carbon dioxide (CO2) is known as a greenhouse gas and plays an essential role in climate change; it is no wonder scientists have been looking for solutions to prevent its release in the environment. However, as a cheap, readily available and non-toxic carbon source, in the past few years there have been efforts to turn carbon dioxide into valuable wares, or ‘value-added’ products.
For instance, carbon dioxide enables energy storage by reacting with hydrogen gas - called the hydrogenation process - transforming the mixture into higher energy liquid compounds such as methanol that can be easily transported and used as fuel for cars. Similarly, carbon dioxide hydrogenation in the presence of other chemicals can lead to the formation of various value-added products widely used in industry such as formic acid, formamides, or formaldehyde. These chemicals can also potentially be used for energy storage as, for example, heating formic acid under certain conditions allow for the release of hydrogen gas in a controlled and reversible fashion. Conversion of carbon dioxide into useful products is complicated by the fact that CO2 is the most oxidized form of carbon and as such a very stable and unreactive molecule. Therefore, the direct reaction of CO2 with hydrogen requires high energy, making the process economically unfavorable. This problem can be overcome using catalysts, which are compounds used in small amounts to accelerate chemicals reactions. For CO2 hydrogenation purposes, most known catalysts are based on precious metals such as iridium, rhodium or ruthenium. While excellent catalysts, the scarcity of these precious metals makes it difficult to use them at industrial scales. They are also hard to recycle and potentially toxic for the environment. Other catalysts use cheaper metals such as iron or cobalt but require a phosphorus-based molecule - called phosphine -surrounding the metal. Phosphines are not always stable around oxygen and sometimes burn violently in an air atmosphere, which presents another problem for the practical applications.
Read more at Okinawa Institute of Science and Technology (OIST) Graduate University
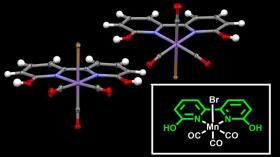
Image: The manganese atom (in purple) is at the center of the frame -- the ligand -- which facilitates the hydrogenation of CO2. (Credit: OIST)