
Resilient bacteria might help streams but could threaten human health
(Millbrook, NY) In urban streams, persistent pharmaceutical pollution can cause aquatic microbial communities to become resistant to drugs. So reports a new study published today in the journal Ecosphere.
Emma Rosi, an aquatic ecologist at the Cary Institute of Ecosystem Studies and lead author on the study explains, "Wastewater treatment facilities are not equipped to remove many pharmaceutical compounds. We were interested in how stream microorganisms - which perform key ecosystem services like removing nutrients and breaking down leaf litter - respond to pharmaceutical pollution."
>> Read the Full Article
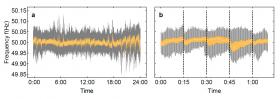
Our power grid works at a frequency of 50 hertz – usually generated by turbines, for example in hydro- or coal power plants, which rotate at a speed of 50 revolutions per second. "When a consumer uses more electrical energy from the power grid, the grid frequency drops slightly before an increased energy feed-in re-establishes the original frequency," explains Benjamin Schäfer from the Max Planck Institute for Dynamics and Self-Organization (MPIDS) in Göttingen and lead author of the study. "Deviations from the nominal value of 50 hertz must be kept to a minimum, as otherwise sensitive electrical devices could be damaged."
>> Read the Full Article
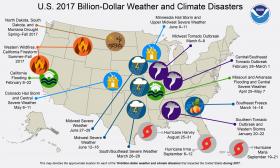
2017 will be remembered as a year of extremes for the U.S. as floods, tornadoes, hurricanes, drought, fires and freezes claimed hundreds of lives and visited economic hardship upon the nation. Recovery from the ravages of three major Atlantic hurricanes making landfall in the U.S. and an extreme and ongoing wildfire season in the West is expected to continue well into the new year.
>> Read the Full Article

A University of Victoria molecular biologist has gained new insights into how environmental contaminants may disrupt thyroid systems. The discovery was made while assembling the genome of the North American bullfrog.
Caren Helbing’s findings could help explain the mechanisms of early development, as well as how environmental contaminants cause thyroidrelated diseases and malfunctions.
>> Read the Full Article

 ENN
Environmental News Network -- Know Your Environment
ENN
Environmental News Network -- Know Your Environment