Engineers at the University of Illinois Chicago have built a cost-effective artificial leaf that can capture carbon dioxide at rates 100 times better than current systems.
Engineers at the University of Illinois Chicago have built a cost-effective artificial leaf that can capture carbon dioxide at rates 100 times better than current systems. Unlike other carbon capture systems, which work in labs with pure carbon dioxide from pressurized tanks, this artificial leaf works in the real world. It captures carbon dioxide from more diluted sources, like air and flue gas produced by coal-fired power plants, and releases it for use as fuel and other materials.
“Our artificial leaf system can be deployed outside the lab, where it has the potential to play a significant role in reducing greenhouse gases in the atmosphere thanks to its high rate of carbon capture, relatively low cost and moderate energy, even when compared to the best lab-based systems,” said Meenesh Singh, assistant professor of chemical engineering in the UIC College of Engineering and corresponding author on the paper.
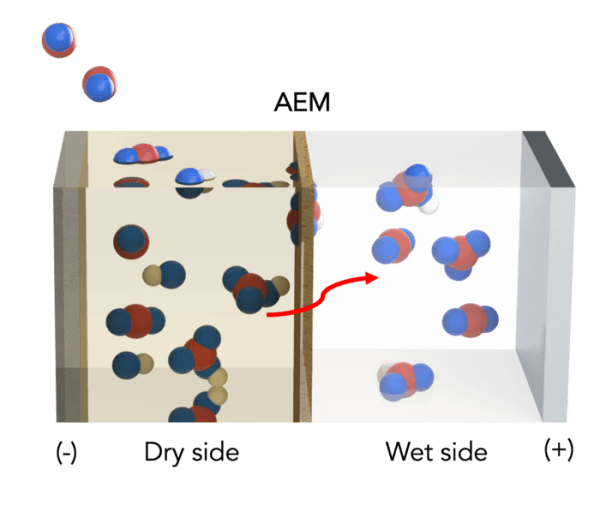
Using a previously reported theoretical concept, the scientists modified a standard artificial leaf system with inexpensive materials to include a water gradient — a dry side and a wet side — across an electrically charged membrane.
On the dry side, an organic solvent attaches to available carbon dioxide to produce a concentration of bicarbonate, or baking soda, on the membrane. As bicarbonate builds, these negatively charged ions are pulled across the membrane toward a positively charged electrode in a water-based solution on the membrane’s wet side. The liquid solution dissolves the bicarbonate back into carbon dioxide, so it can be released and harnessed for fuel or other uses.
Read more at University of Illinois Chicago
Image: Illustration of a carbon capture process designed by UIC College of Engineering scientists. Carbon dioxide from air or flue gas is absorbed by a dry organic solution to form bicarbonate ions, which migrate across a membrane and are dissolved in a liquid solution to concentrated CO2. Carbon atoms are shown in red, oxygen atoms are shown in blue and hydrogen atoms are shown in white. (Credit: Aditya Prajapati/UIC)